The “FDA approved version” of the Pfizer-BioNTech vaccine has been [legally] available since Aug. 23, 2021, however the new version of the popular inoculation, is still not available in America. Pfizer-BioNTech works to rebrand under the new name “COMIRNATY”, allowing the now legal injection to be marketed, and mandated, to the public.
Comirnaty vs Pfizer-BioNTech?
The FDA’s website states, “The FDA-approved Comirnaty (COVID-19 Vaccine, mRNA), made by Pfizer for BioNTech and the FDA-authorized Pfizer-BioNTech COVID-19 Vaccine under EUA have the same formulation and can be used interchangeably to provide the COVID-19 vaccination series without presenting any safety or effectiveness concerns.”
When the FDA grants full approval, it means “the agency has determined that the benefits of the product outweigh the known risks for the intended use.” as stated on the FDA’s website. Before this approval, the coronavirus vaccinations were released using emergency use authorization.
Legality of Emergency Use Authorization
The emergency use authorization version of Pfizer-BioNTech’s coronavirus concoctions may be the same as COMIRNATY, however legally they are completely different entities. This means, according to the Epoch Times, that the government cannot force civilians, nor military, to inject these experimental bottled versions. The new labeled [legally approved] version is expected to be available to local pharmacies by early November 2021.

“You cannot interchange BioNTech’s Cominarty with Pfizer’s [shot] from a legal standpoint. They are legally distinct” Matthew Staver, chairman of Christian legal group Liberty Counsel stated to the Epoch Times
According to numerous Departments of Health, despite the mandate for the FDA approved version of the drug, Comirnaty, still hasn’t even hit shelves in America.
When will COMIRNATY be available?

“The CDC anticipates that Comirnaty will begin to ship some time in November at the earliest,” a spokesman for the Arizona Department of Health told The Epoch Times in an email, referring to the Centers for Disease Control and Prevention
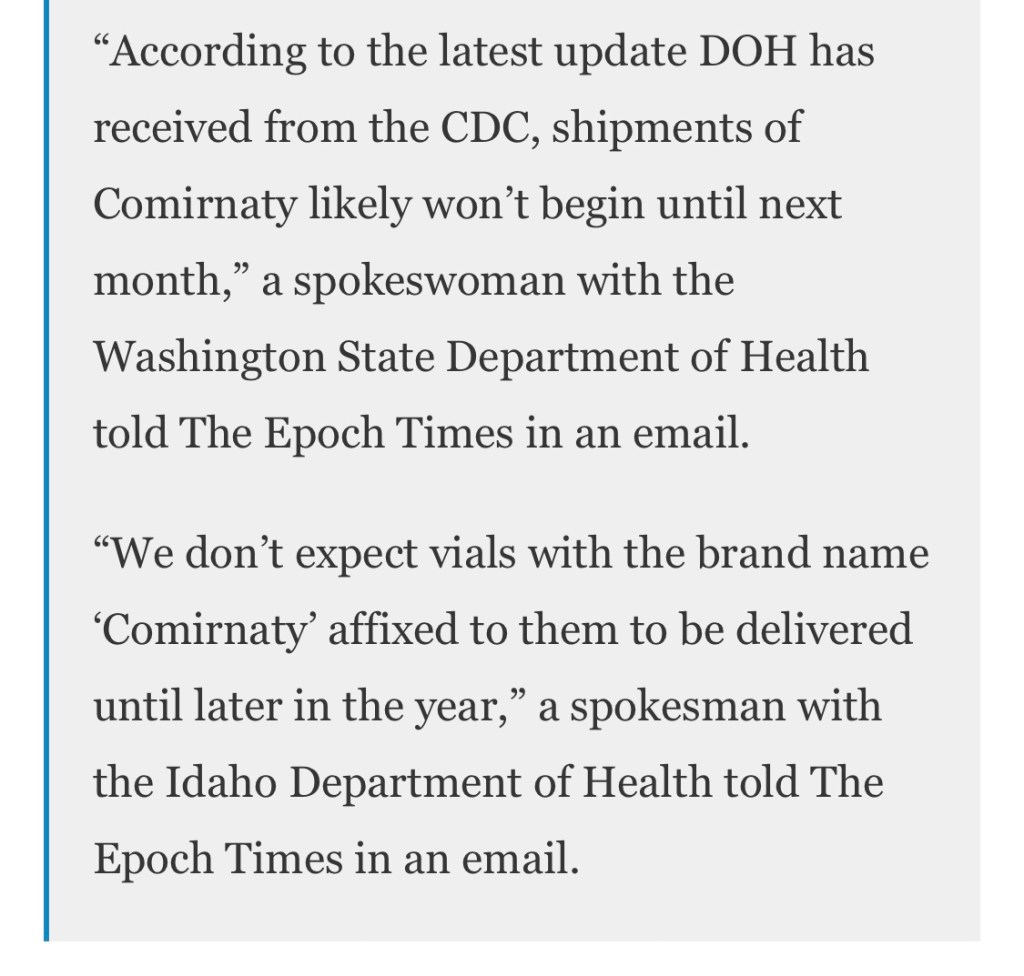
“We don’t expect vials with the brand to name ‘Comirnaty’ affixed to them to be delivered until later in the year,” a spokesman with the Idaho Department of Health told The Epoch Times in an email
“The Maryland Department of Health has been told by the CDC that Comirnaty will not ship until the end of October/beginning of November,” a spokesman with the Maryland Department of Health told The Epoch Times in an email.
“According to the latest update DOH has received from the CDC, shipments of Comirnaty likely won’t begin until next month,” a spokeswoman with the Washington State Department of Health told The Epoch Times in an email.
Conclusion
America is responding to an emergency use authorization vaccine as if it were the FDA approved version. This has allowed federal and state mandates that continue to ravage small businesses, and discriminate against all minorities.
Should America continue to treat their Emergency Use Authorize version of the same vaccine as the legally approved version? If following the technicality of our U.S. laws are not important, why does American laws exist in the first place? Should major pharmaceutical manufacturers, their stakeholders, and the FDA be allowed to overlook laws, while the citizens are demonized for opposing them?
Update –
- As of November 12th, 2021, the “approved” version of the Comirnaty vaccine is still not available in the United States.

