The U.S. Food and Drug Administration’s “vaccine advisory panel” on Friday voted in a landslide 16-3 against providing mandatory third injections for individuals aged 16 and up. This follows the approval of a booster for those aged 65 and older and for those who are at high risk.
Similar to Israel’s mandatory booster vaccinations, the FDA voted in favor for people aged 65 and up, and those at high risk, booster doses were recommended in an 18-0 vote.
First approved for testing on high risk patients in August, now extends to all Americans over 65. This new approval further desensitizes society to getting routine concoctions every few months. With altered ingredients and increased strength, the boosters—designed to fight the variants—will include side effects potentially more severe than the second dose. No long term studies have been done on the effects of consistent mRNA dosing, and manipulating the body’s messenger system.

Booster Demographic

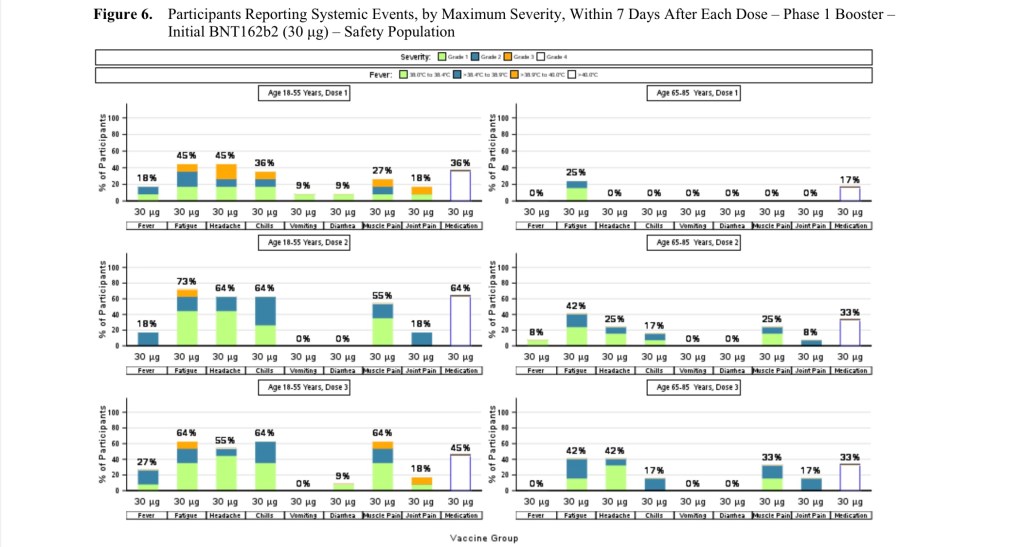
Out of the 306 participants in the trials, 249 of them were white. That means 81.37% of all the participants were white. Why did the clinical trials feel the need to avoid thorough testing of other races? In the confidential document below you can see the statistics in the clinical trials that the FDA have base this approval on.

Could Big Pharmaceutical companies and vaccine mandates be discriminating against American minorities? Read more

