Moderna describes their mRNA platform as the next evolution in modern medicine, improving the discovery, development, and manufacturing of future concoctions in order to combat disease. Moderna states their mRNA technology functions much like a computer operating system. The Pharmaceuatical company describes storage, software, and applications happening inside of the host.
Moderna discusses their technology using forward thinking statements, such as “if,” “should,” and “could,” indicating the current mRNA platform is not its final stage. Moderna feels because of mRNA’s success with COVID-19 that it should work for all disease, eradicating the majority of human suffering from the planet. While this utopian goal exists far in the future, what iterations could be implemented during its progressive evolution?
Moderna describes itself as a “a company committed to innovation1”, who has invested “billions of dollars” into both research and development of mRNA technology. Moderna chose not to enforce patents for their mRNA technology during the pandemic, but warned that post pandemic would require liscensing in order to use their claimed mRNA technology.
The following is a breakdown of Moderna’s self-defined mRNA Platform2:
Moderna’s mRNA Platform
Moderna’s mRNA was developed and released without knowledge of the full extent of the possibilities and its potential long-term side effects. The company states they built their entire company under the premonition that if mRNA “works for one disease it should work for many diseases.” This ludicrous assumption has brought Moderna to push their mRNA technology as an essential component to our own vitality, including it in the mandates, allowing mRNA to be injected into the arms of both willing and unwilling individuals across the nation.
“We built Moderna on the guiding premise that if using mRNA as a medicine works for one disease, it should work for many diseases. And, if this is possible – given the right approach and infrastructure – it could meaningfully improve how medicines are discovered, developed and manufactured.”
Operating System
Moderna states that they have created an “mRNA platform”, which they claim functions as an operating system. Using “plug and play” technology, or easily swappable components, mRNA technology is stated to be able to manipulate cell production of proteins. Moderna refers to this technology as an “app” or a “program”.
“Our Operating System”
“Recognizing the broad potential of mRNA science, we set out to create an mRNA technology platform that functions very much like an operating system on a computer. It is designed so that it can plug and play interchangeably with different programs. In our case, the ‘program’ or ‘app’ is our mRNA drug – the unique mRNA sequence that codes for a protein.”
“We have a dedicated team of several hundred scientists and engineers solely focused on advancing Moderna’s platform technology. They are organized around key disciplines and work in an integrated fashion to advance knowledge surrounding mRNA science and solve for challenges that are unique to mRNA drug development. Some of these disciplines include mRNA biology, chemistry, formulation & delivery, bioinformatics and protein engineering.”
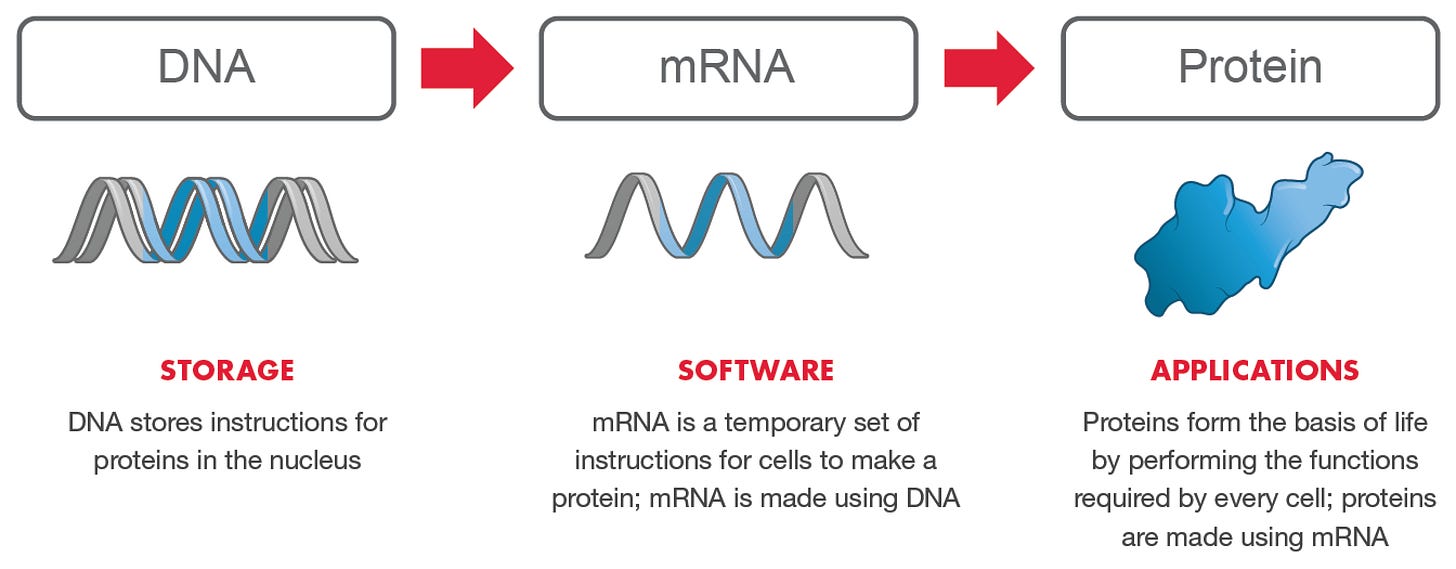
The Software of Life
Moderna considers their mRNA technology to be “the software of life.” The mRNA is “coded” with “genetic instructions” using “investigational mRNA” to provide the host with a “software-like quality.” The only difference between Moderna’s mRNAs is the genetic instructions that causes ribosomes to produce a protein. These proteins are responsible for creating an immunoresponse which produces an immunity to the disease. Moderna’s mRNA provides the ability to use a template for the rapid development of new genetic instructions (code) which can be added to, forming innovative vaccines and treatments.
Now scientists have the ability to manipulate human life, extending the survival of our species. The medical community, working in conjunction with governments around the world, has forcibly implemented mRNA technology in order to reap profits, promoting a narrow view of acceptable treatment options. During the next decade, Moderna and their shareholders aim to convert many traditional vaccines into mRNA substitutes.
“Our mRNA Medicines – The ‘Software of Life’”
“When we have a concept for a new mRNA medicine and begin research, fundamental components are already in place.”
“Generally, the only thing that changes from one potential mRNA medicine to another is the coding region – the actual genetic code that instructs ribosomes to make protein. Utilizing these instruction sets gives our investigational mRNA medicines a software-like quality. We also have the ability to combine different mRNA sequences encoding for different proteins in a single mRNA investigational medicine.”
“We are leveraging the flexibility afforded by our platform and the fundamental role mRNA plays in protein synthesis to pursue mRNA medicines for a broad spectrum of diseases.”
mRNA DESIGN STUDIO
To materialize this notion, Moderna has named their research engine the “SOFTWARE OF LIFE”, which includes an “mRNA DESIGN STUDIO.” Genetic modification will soon become ubiquitous to modern medicine, shaping the fingerprint of our species, giving the iatrarchy complete God-like control over humanity. In time, the software of life may have the ability to completely re-write our biological structure to our preference, eventually becoming an completely artificial immune system, our sustainability fully reliant on the medical dictatorship.
“As our scientists create new mRNA concepts, they can design mRNAs for research and testing, within days, using our proprietary systems.”
“As the DIGITAL BIOTECH COMPANY™, we utilize the software-like property of mRNA in our proprietary, web-based mRNA DESIGN STUDIO.”
“Our scientists request mRNAs for a specific protein, and the protein target is automatically converted to an initial optimized mRNA sequence. Using our Sequence Designer module, they can tailor entire mRNAs from the 5’-UTR to the coding region to the 3’-UTR based on our ever-improving proprietary learnings. The mRNA sequence is then further optimized using our proprietary bioinformatics algorithms.”
“Our digital ordering then ensures rapid and accurate transmission of sequences to our modular synthesis robotics.”
“Scientists can begin by selecting any protein in the human proteome to be further engineered, including antibodies, or they can design novel proteins like traps, fusion proteins, or completely novel scaffolds and sequences. All can be designed to explore previously undruggable pathways.”
mRNA Coding
Moderna displays a diagram on their website, showing the Coding Region. This is where the mRNA instructions are written. Each booster shot functions as an update to the program, allowing new instructions to be sent out, utilizing the same basic mRNA platform to execute these instructions.
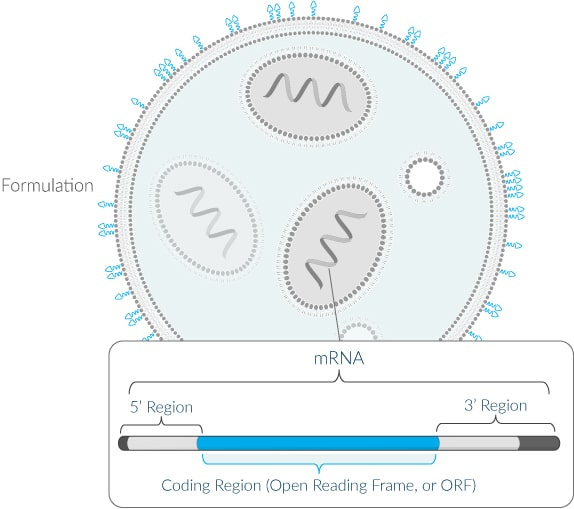
“Within a given modality, the base components are generally identical across development candidates – formulation, 5’ region and 3’ region. Only the coding region varies based on the protein/s the potential medicine is directing cells to produce.”
“Learn how our Research Engine and Early Development Engine are enabling us to fully maximize the promise of mRNA to meaningfully improve how medicines are discovered, developed and manufactured.”
The future of mRNA allows the possibility for revolutionary medicine in ways previously impossible, however with these advances come setbacks, obstacles, and concerns. One of these concerns are the long-term effects of cell manipulation through consistent mRNA injections. Another harrowing perspective are the concoctions blend of inactive ingredients, such as PEG, in addition to use of synthetic lipid nanoparticles for the delivery of mRNA.
The Future of Moderna’s mRNA
With mRNA, scientists have the ability to rapidly develop new drugs in a fraction of the time previously required, allowing the possibility to treat existing diseases, alongside newly emerging ones. With the warp speed discovery of new mRNA concoctions, comes the possibility for error, as these products can be brought to market, and even mandated on the populous in a short period of time. With mRNA vaccines, long-term effects are determined in theory, as seen with the COVID-19 vaccine. This can present future unintended consequences, however Moderna remains forward thinking in the long-term implementation of this technology.
“We aim to accelerate the pace and success rate of mRNA discovery research efforts across our therapeutic areas and for our strategic collaborators.”
“With the ability to receive high-quality mRNA constructs in a short period of time, scientists are able to quickly run experiments to test a concept.”
“Additionally, scientists can design large parallel experiments with multiple comparison arms using different mRNA constructs.”
“This enables scientists to narrow their field of drugs candidates rapidly, ultimately bringing forth development candidates that can advance into and through the clinical development process.”
“Overcoming Key Challenges”
“Using mRNA to create medicines is a complex undertaking and requires overcoming novel scientific and technical challenges. We need to get the mRNA into the targeted tissue and cells while evading the immune system. If the immune system is triggered, the resultant response may limit protein production and, thus, limit the therapeutic benefit of mRNA medicines. We also need ribosomes to think the mRNA was produced naturally, so they can accurately read the instructions to produce the right protein. And we need to ensure the cells express enough of the protein to have the desired therapeutic effect.”
“Our multidisciplinary platform teams work together closely to address these scientific and technical challenges. This intensive cross-functional collaboration has enabled us to advance key aspects of our platform and make significant strides to deliver mRNA medicines for patients.”
According to Moderna, the company “has been granted more than 350 patents in the United States, Europe, Japan and other jurisdictions, protecting fundamental inventions in the mRNA therapeutics space, with several hundred additional pending patent applications covering key advances in the field”
While Moderna does not publicly avow of biosensor technology in development, in 2013 DARPA awarded Moderna $25 million for the research and develop of mRNA technology3. This includes aerosol monitoring for SARS-CoV-2 using their Resource Effective Bio-identification System (REBS). In 2011 DARPA began investigating the development of nucleic acid vaccines. In December 2020, a former ADEPT mRNA vaccine developed by Moderna received the green light for mass injections.
DARPA
In 2016, DARPA initiated the Pandemic Prevention Platform (P3) for the rapid development of antibodies to combat future emerging diseases. Under this platform, scientists were able to successfully ‘find and manufacture antibodies in ninety days, when previous methods have traditionally take years. This research shifted focus through the emergence of SARS-CoV-2 to the development of a successful mRNA COVID-19 vaccine.
“In October 2013, DARPA awarded Moderna up to approximately $25 million to research and develop potential mRNA medicines as a part of DARPA’s Autonomous Diagnostics to Enable Prevention and Therapeutics, or ADEPT, program, which is focused on assisting with the development of technologies to rapidly identify and respond to threats posed by natural and engineered diseases and toxins. This award followed an initial award from DARPA given in March 2013.”
“As part of the ADEPT program in 2011, DARPA began investing in nucleic acid vaccines. The hypothesis was that rather than delivering antigens to the immune system, we [DARPA] could deliver genes that encode the antigen and allow the human body to produce the antigen from its own cells, triggering a protective immune response. In December 2020, former ADEPT performer Moderna’s RNA vaccine received FDA Emergency Use Authorization (EUA) approval for the prevention of COVID-19.”
“In FY2016, DARPA initiated the Pandemic Prevention Platform (P3) program aimed squarely at the rapid discovery, testing, and manufacture of antibody treatments to fight any emerging disease threat. P3 convincingly demonstrated how to find and manufacture antibodies in less than 90 days (vs. years), using influenza, Zika, and MERS as test cases. As the COVID-19 outbreak began early in 2020, P3 research pivoted to address the novel coronavirus.”
According to DARPA, researchers at Standford University are currently in development of biosensors that will be able to detect a COVID-19 invasion, through cell membrane surveillance. As this technology replaces existing testing devices, future iterations could begin to use implanted biosensors to detect potential attacks on cell membranes. Could Stanford’s biosensor research, and similar implanted contraptions, be integral in Moderna’s mRNA platform?
“Researchers at Stanford University are developing biosensors that can quickly spot an attack on a cell membrane – the first step of CoVID-19 (or any) infection. The team is using the platform to identify mechanisms to inhibit CoVID-19 membrane attack; the technology can be readily multiplexed enabling fast high-throughput drug screens.”
Biosensors, 5G, Microneedle Patches, and Beyond
If mandatory implanted biosensors to track COVID-19 and produce consistent biological metadata sounds like science fiction, consider iterations of these plans have already been in effect from oligarchs and organizations for years.
On Apr 28, 2016, Sundar Pichai, CEO of Google and Alphabet, warned “Looking to the future, the next big step will be for the very concept of the ‘device’ to fade away4.” Although Pichai’s words were not targeting COVID-19 tests, it seems inevitable that medical device manufactures will adopt the same technology as existing consumer electronics. This means, as the cellular device becomes one with our biological structure, so too will medical surveillance devices, to ensure mass compliance with public health demands.
The World Economic Forum
On January 10th, 2016 Klaus Schwab of the World Economic Forum publicly predicted there would be implantable microchips in an interview “Certainly in the next ten years.” These views were also written in his books, including “The Fourth Industrial Revolution.” Schwab added, “And at first we will implant them in our clothes. And then we could imagine that we will implant them in our brains, or in our skin. And in the end, there will be a direct communication between our brain and the digital world. What we see is a kind of fusion of the physical, digital and biological world.” Klaus Schwab is an influential thinker throughout the philanthropic, scientific, and medical community.
The FDA
On November 13th, 2017, the U.S. Food and Drug Administration approved the first electronic pill that allows the tracking of the ingested medication5. This ensures the patient complies and takes the pill.
“The U.S. Food and Drug Administration today approved the first drug in the U.S. with a digital ingestion tracking system. Abilify MyCite (aripiprazole tablets with sensor) has an ingestible sensor embedded in the pill that records that the medication was taken. The product is approved for the treatment of schizophrenia, acute treatment of manic and mixed episodes associated with bipolar I disorder and for use as an add-on treatment for depression in adults.”
Pfizer
While an electronic pill to help patients may sound innocuous, on January 24th, 2018 Pfizer CEO Albert Bourla spoke at the World Economic Forum praising the release of the trackable medical product, which Bourla recognized as a tool of “compliance.”
Q: “Question about patient engagement…[E]ven if you make the greatest drug, or the greatest wearable, there’s no guarantee that the patient will take the drugs or wear the device. So how are you thinking about technology to engage the patient?”
Albert Bourla: “Yeah, again, maybe I will use an example, I think it’s fascinating what’s happening in this field right now. I mean, FDA approved the first electronic pill, if I can call it like that. So, it is basically biological. chip that it is in the tablet. And once you take the tablet and dissolves into your stomach, sends a signal that you do the tablet. So imagine the applications of that: Compliance. The insurance companies to know that the medicines that patients should take they do take them. It is fascinating what happens in this field. But of course, there will be an initial cost, that someone needs to invest”
Could Moderna implement a digital ingestion tracking system alongside their mRNA vaccines? This would eliminate the need for vaccine passports, and taking time to checking the status of the vaccinated. It would also allow those who are unvaccinated to be easily recognized, and handled accordingly.
This technology can be considered essential in many instances, such as the “electronic pill” for patients who truly require digital assistance reminders with their daily medical requirements. As it becomes more widely available, it can also be turned against the populous, producing a totalitarian state of medical enforcement.
Microsoft
On March 26, 2020 Microsoft published patents titled “WO2020060606 – CRYPTOCURRENCY SYSTEM USING BODY ACTIVITY DATA6” and “US20200097951 – CRYPTOCURRENCY SYSTEM USING BODY ACTIVITY DATA7” which uses body activity data to verify access to cryptocurrency network and systems.
If body activated cryptocurrency systems makes its way to the mainstream market, it could be just one of many surveillance contraptions imposed on humanity. These patents show the trend and direction of future technologies. As they diminish in size, devices will become more invasive, allowing our biological metadata to influence our environment.
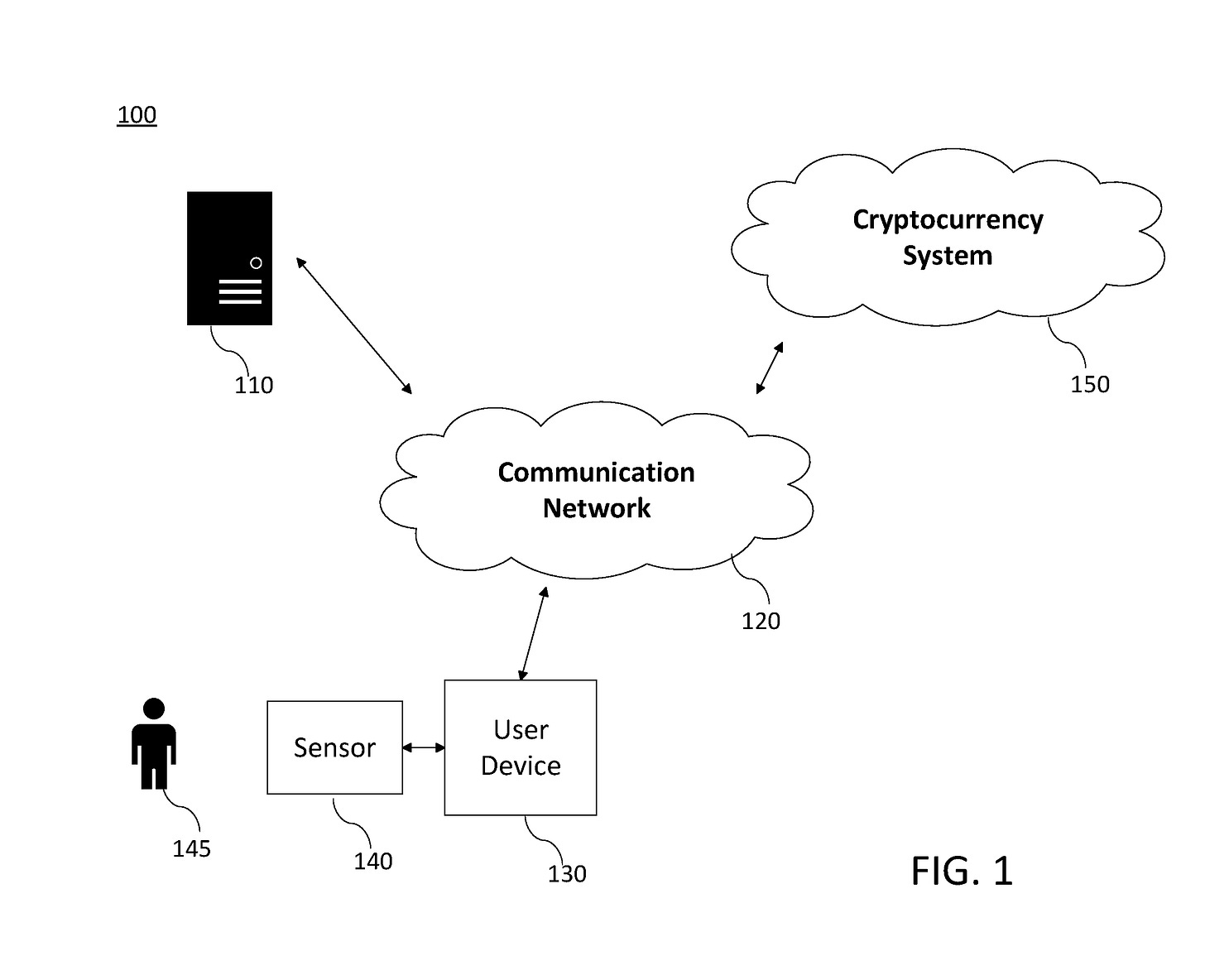
“Human body activity associated with a task provided to a user may be used in a mining process of a cryptocurrency system. A server may provide a task to a device of a user which is communicatively coupled to the server.”
“A sensor communicatively coupled to or comprised in the device of the user may sense body activity of the user. Body activity data may be generated based on the sensed body activity of the user.”
“The cryptocurrency system communicatively coupled to the device of the user may verify if the body activity data satisfies one or more conditions set by the cryptocurrency system, and award cryptocurrency to the user whose body activity data is verified.”
Dissolvable Microneedle Patches
Dissolvable Microneedle Patches may be the next prophylactic mRNA treatment for SARS-CoV-2. Human trials for health-care worker administered and self-administered dissolvable microneedle patches began June 27th, 2017, funded by the National Institutes of Health8 (NIH).
Three of the researchers who conducted the studies were inventors on licensed patents and have ownership interest in companies developing microneedle products for Micron Biomedical. Conflicts of interest are typical in clinical trials, which allow many treatments to pass through FDA regulations, despite their potential unknown effects when unleashed upon the diverse patients of the world. Conflicts of interest allow companies to make a faster profit, as the researchers have predetermined beliefs that the treatment works.
“Microneedle patches provide an alternative to conventional needle-and-syringe immunisation, and potentially offer improved immunogenicity, simplicity, cost-effectiveness, acceptability, and safety. We describe safety, immunogenicity, and acceptability of the first-in-man study on single, dissolvable microneedle patch vaccination against influenza.”
“Use of dissolvable microneedle patches for influenza vaccination was well tolerated and generated robust antibody responses.”
Rouphael NG, Paine M, Mosley R, Henry S, McAllister DV, Kalluri H, Pewin W, Frew PM, Yu T, Thornburg NJ, Kabbani S, Lai L, Vassilieva EV, Skountzou I, Compans RW, Mulligan MJ, Prausnitz MR; TIV-MNP 2015 Study Group. The safety, immunogenicity, and acceptability of inactivated influenza vaccine delivered by microneedle patch (TIV-MNP 2015): a randomised, partly blinded, placebo-controlled, phase 1 trial. Lancet. 2017 Aug 12;390(10095):649-658. doi: 10.1016/S0140-6736(17)30575-5. Epub 2017 Jun 27. PMID: 28666680; PMCID: PMC5578828.
Conclusion
While many of these technologies are not immediately available solutions, their existence is inevitable. Invasive medical technology is unlikely to begin its societal implementation as totalitarian tactics of control, however as the world saw with SARS-CoV-2 vaccine mandates, if the demand is right—potential for profit—anything is possible. The mass resistance to innovative medical technology and their shareholders resulted in authoritative forced compliance and the revocation of civil rights.
“You dont want to wait in line to get a COVID-19 test? Get a medical implant today and have rapid real-time viral detection at a glance”
“Get ahead of the curve: Order your dissolvable microneedle vaccines today, and save time this season. Family discounts available.”
The above phrases may be the campaigns from the future devices pushed by the ascending global iatrarchy. With mandates and forced compliance, come “choices” which can be positioned in a way to offer the incentive of regaining freedom, an illusion depicting to influence persuasion.
Moderna’s mRNA may be “built to function like an operating system”, using booster injections for perpetual upgrades, but how will this wording change with the evolution of innovative medical products. With technological advances come heightened concern for regulatory long-term safety testing—in correlation with human dependence once this technology is introduced into society. Moderna may be normalizing this terminology as a precursor for the next step in their strategic plan to rollout implanted biosensors, and dissolvable microneedle patches using mRNA technology as ‘“software updates”, rolling out in conjunction with the emergence of new variants and hazards. Could the development of 5G technology ultimately play a role in the advancement of real-time testing devices, biosensors, and the eventual automatic administering of mRNA vaccines? Is Moderna’s description of their mRNA technology functioning as an “operating system” an insight of their long-term goals to infuse medicine and technology? Or, this could simply be a poor choice of words conducted by Moderna, in an attempt to explain their mRNA technology to the public?
Freedom of speech, freedom of press, and freedom of thought in America allows the possibilities for both ideas to be discussed.