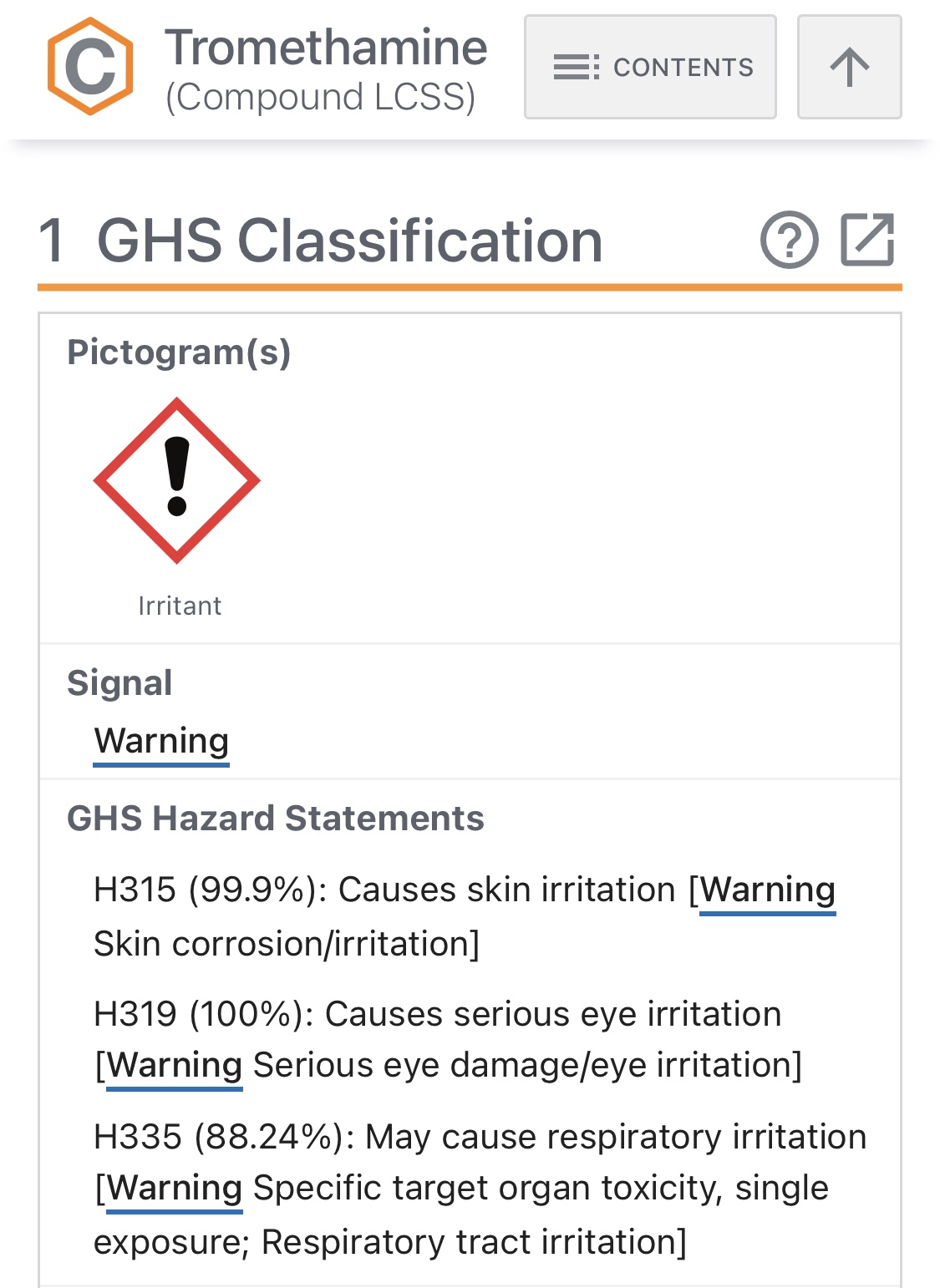
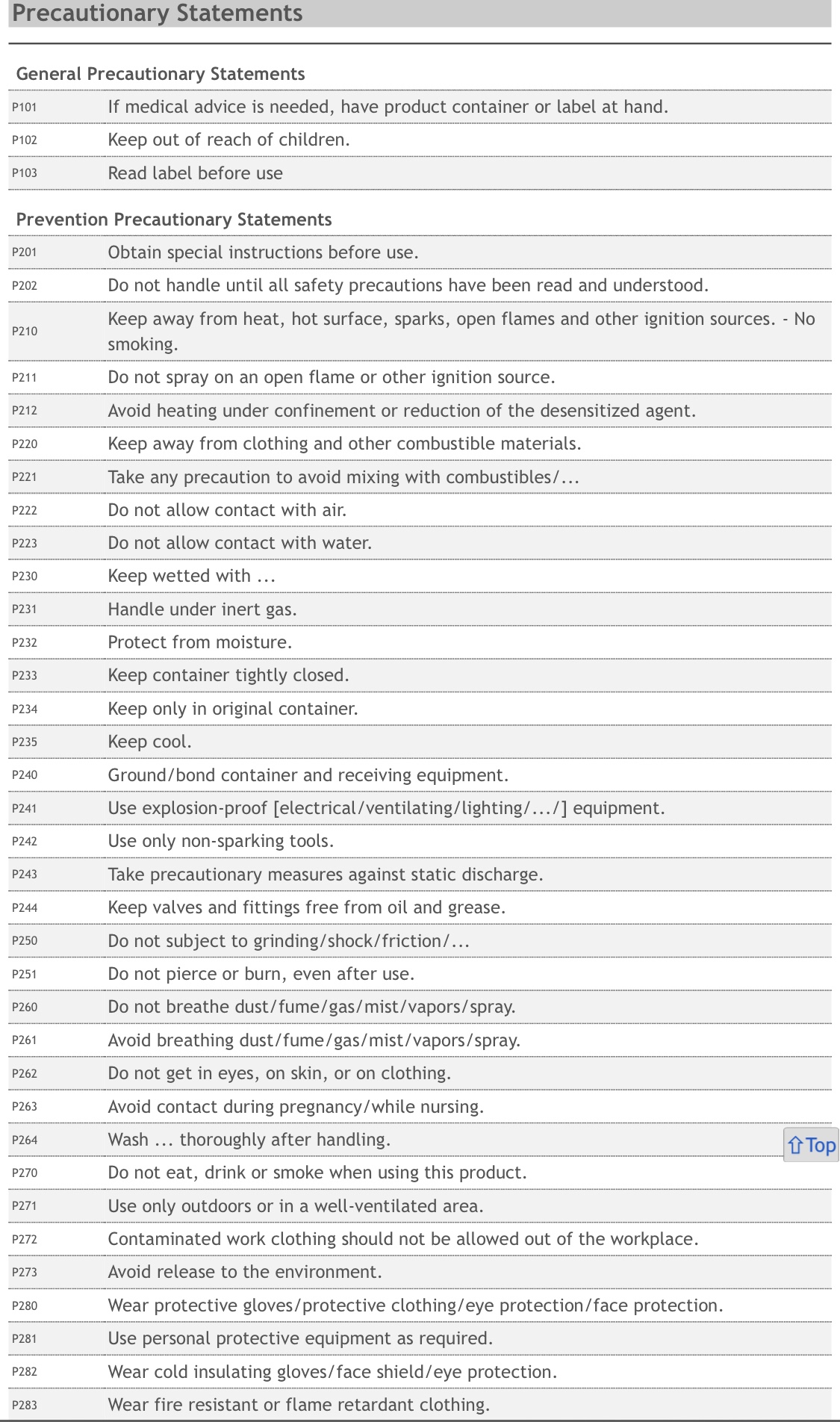
Should a potentially toxic chemical used inside coronavirus injections, be mandated for children aged five to eleven?
On October 29th, 2021 the U.S. Food and Drug Administration’s (FDA) ‘vaccine advisory panel’ passed a unanimous vote (17-0), recommending the FDA officially authorize the “emergency” use of Pfizer-BioNTech’s coronavirus concoctions for use in children aged 5 – 11 years.
Pfizer’s safety data comes not only from their experimental trials which tested potential harmful vaccine effects on 2,268 “volunteered” children, but from more than 4,600 participants, monitored for safety at least two months after receiving the concoction.

What was not publicized by the unveiling of this new vaccine approval for America’s youth, was the quiet adjustment to the vaccine’s “formula” by Pfizer.
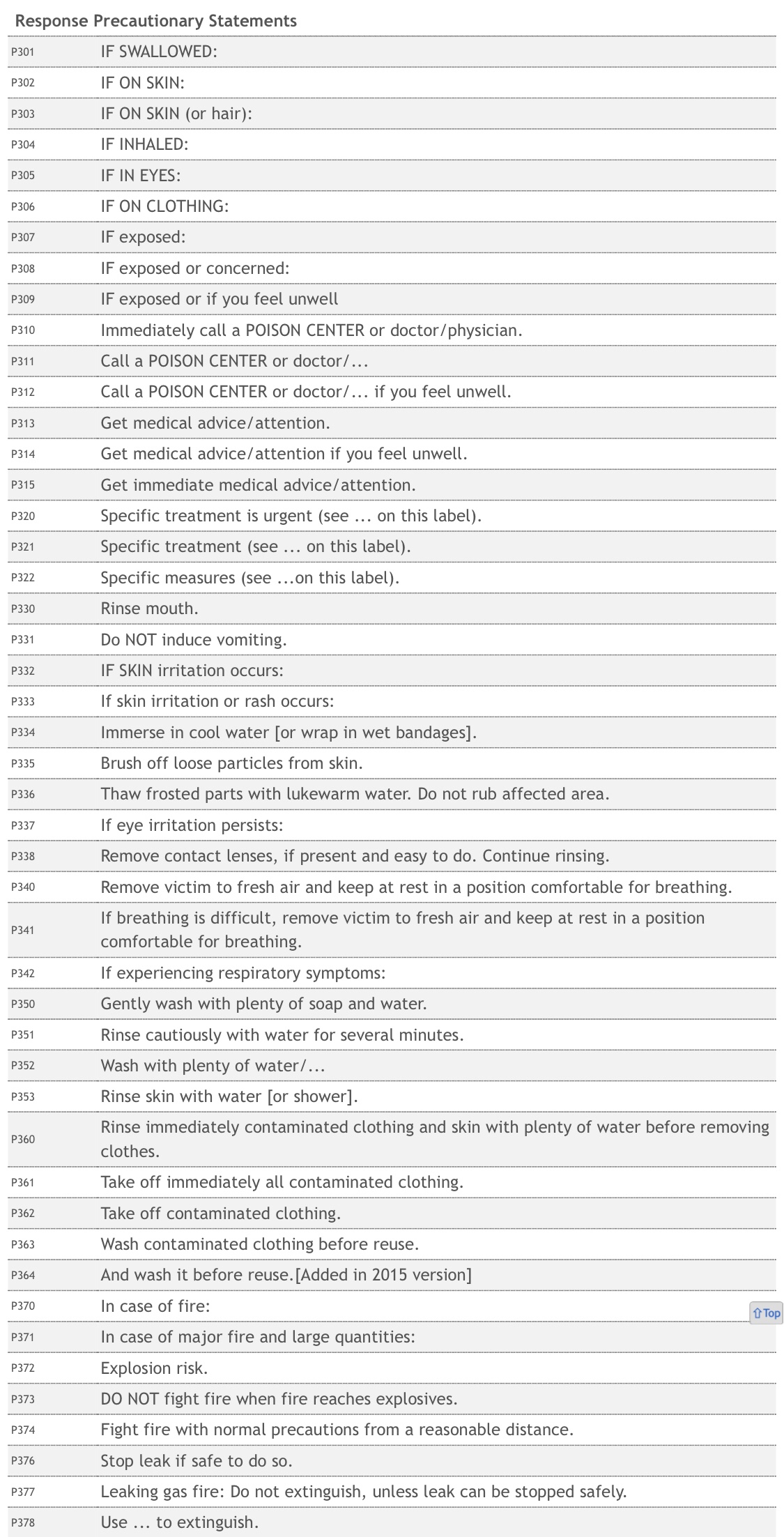
Comirnaty Ingredients –

Taking a closer look at the ingredients of the Comirnaty coronavirus vaccine now injected into children, the pharmaceutical companies Pfizer-BioNTech have altered the vaccine ingredients, for greater efficacy in children aged 5 – 11.
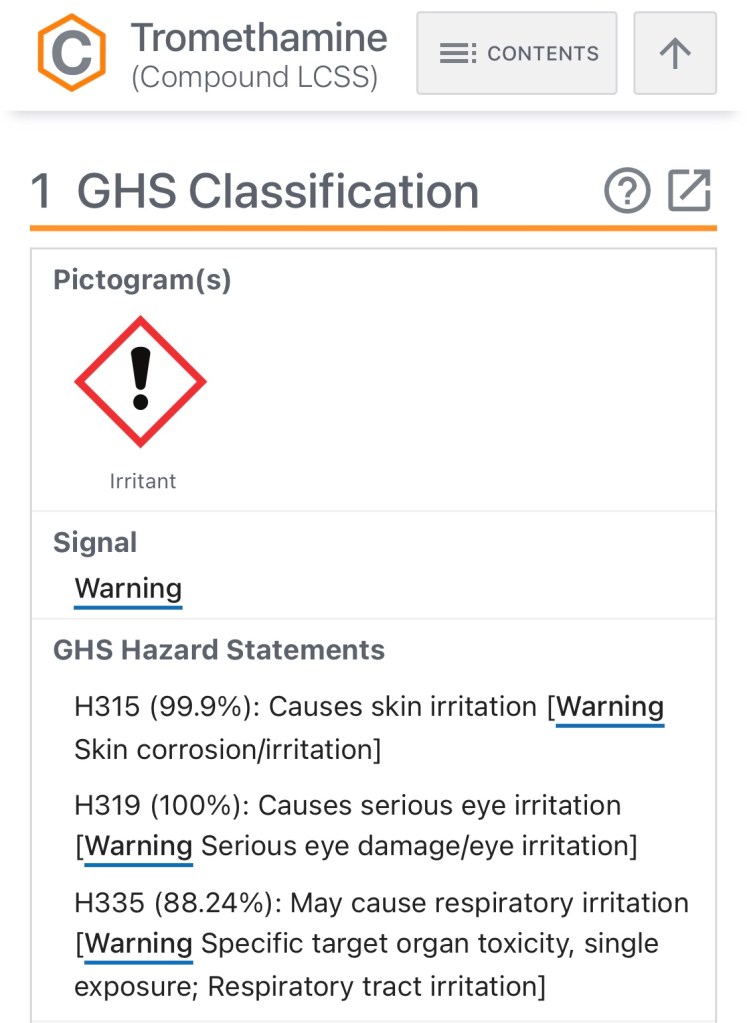
Both vaccines use predominantly identical ingredients. The striking difference between the two Comirnaty injections (adult vs child) is that Pfizer-BioNTech has added a new inactive ingredient to their concoction. The new vaccine formula uses a small dose of a volatile and toxic chemical known as “tromethamine”. Clinical studies show larger amounts of tromethamine is known to cause organ damage, as well as numerous other drug induced injuries with only a single exposure.

Vaccine Formulation
The inactive ingredient tromethamine acts as a buffer, instead of phosphate-potassium chloride (PBS), also excluding sodium chloride and potassium chloride.

Why was this ingredient altered?
The ingredient was altered in order to allow greater stability of the mRNA. Arriving frozen between -130°F/-90°C to -76°F/-60°C the vials are then thawed and refrigerated for up to 10 weeks.

National Institutes of Health
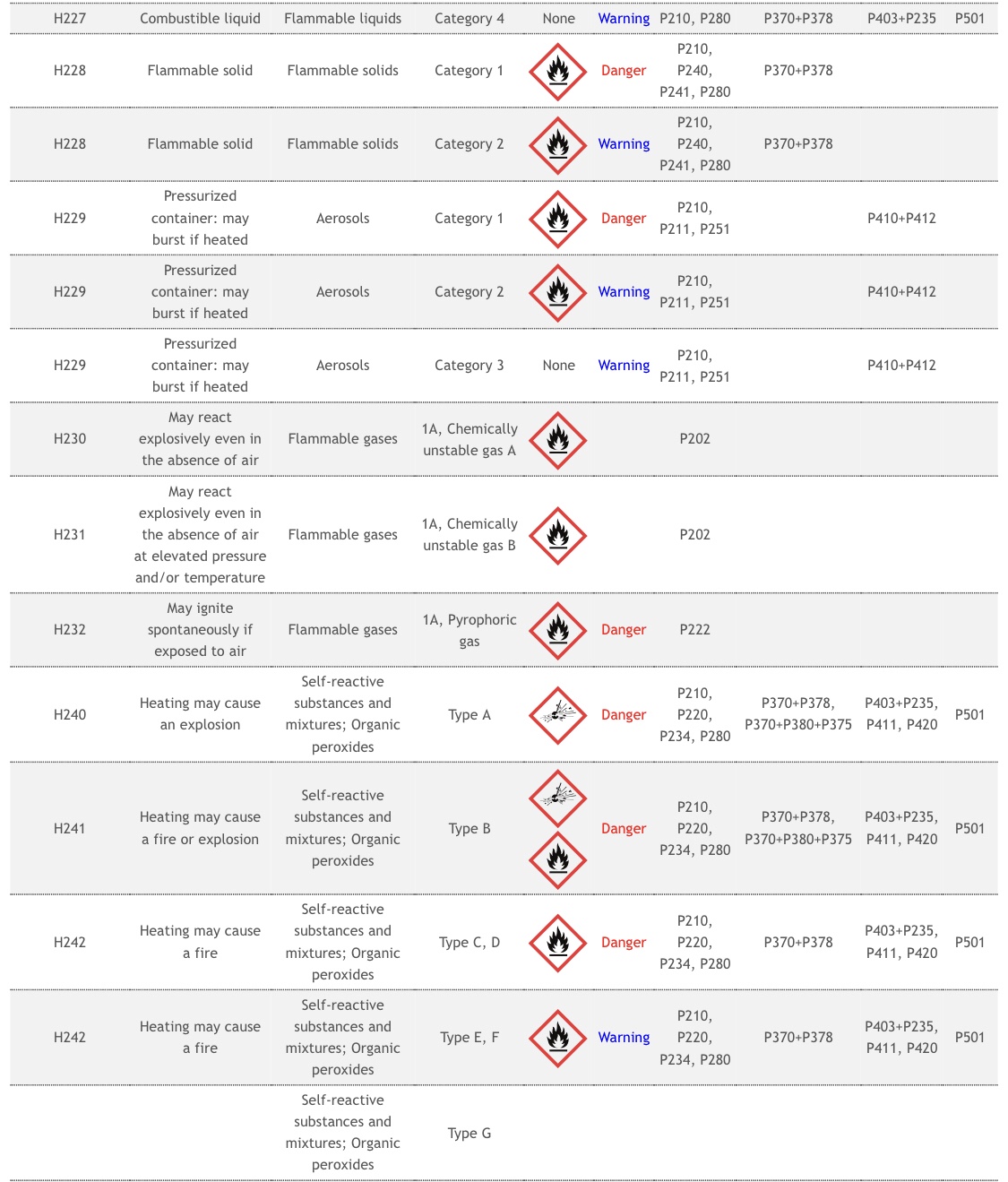
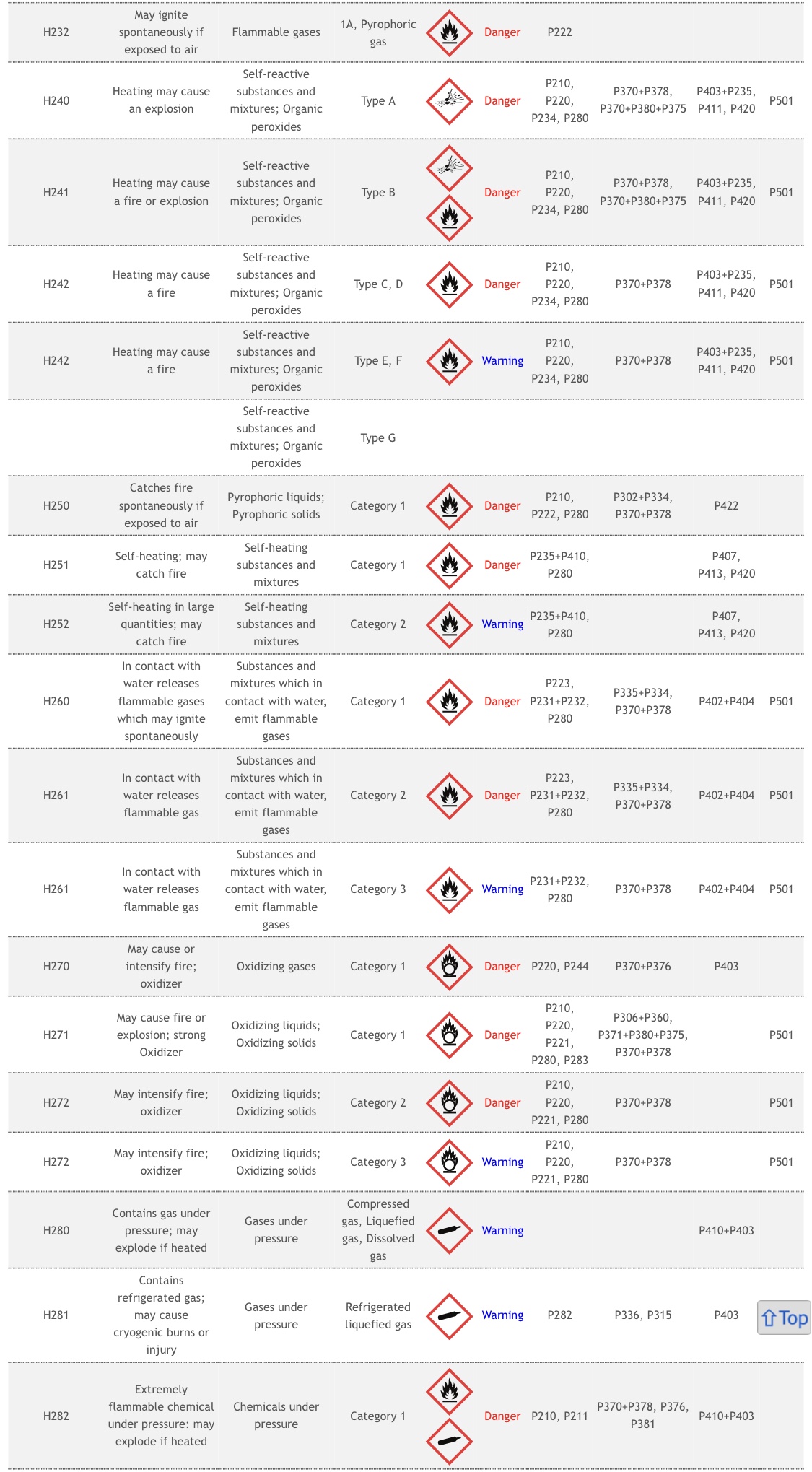
According to the National Institutes of Health, Tromethamine is a toxic and unstable compound that poses significant health risks with just a single exposure.
Although the substance exists in other vaccinations by the FDA, what are the potential risks associated with Tromethamine?
Risks of Tromethamine include –
- Organ Toxicity
- Drug Induced Liver Injury
- Acute Toxicity
- Coma
- Respiratory depression
- Local irritation
- Tissue inflammation,
- Injection site infection
- Febrile response
- Chemical phlebitis
- Venospasm
- Hypervolemia
- IV thrombosis
- Extravasation (with possible necrosis and sloughing of tissues)
- Transient decreases in blood glucose concentrations
- Hypoglycemia
- Hepatocellular necrosis with infusion via low-lying umbilical venous catheters.

Pregnancy Data
Tromethamine is still an under researched chemical substance. it is not known whether tromethamine is distributed in human milk. The National Institutes of Health states “Because many drugs are distributed into milk, the manufacturer recommends the drug be used with caution in nursing women.”


Coronavirus vaccines only contain a small amount of the potentially toxic substance tromethamine. While this chemical does not exist in vaccines suggested for adults, the CDC suggests pregnant women should receive the coronavirus vaccine.
Animal Trials
In Animal trials, rats injected with the substance experienced weakness, collapse, and coma, “even after neutralization”. Injections of high doses [in animals] produced hypoglycemia. Could these reactions be similarly seen in human subjects? It is unknown how long term regular exposure to the substance could effect children aged five to eleven.

The study to test tromethamine toxicity used rats, which have an ~85% genomic similarity as humans. This enables clinical researchers to better predict what will happen upon injection into human beings. Yet many of the side effects were overlooked upon addition of tromethamine to Comirnaty’s coronavirus vaccine.
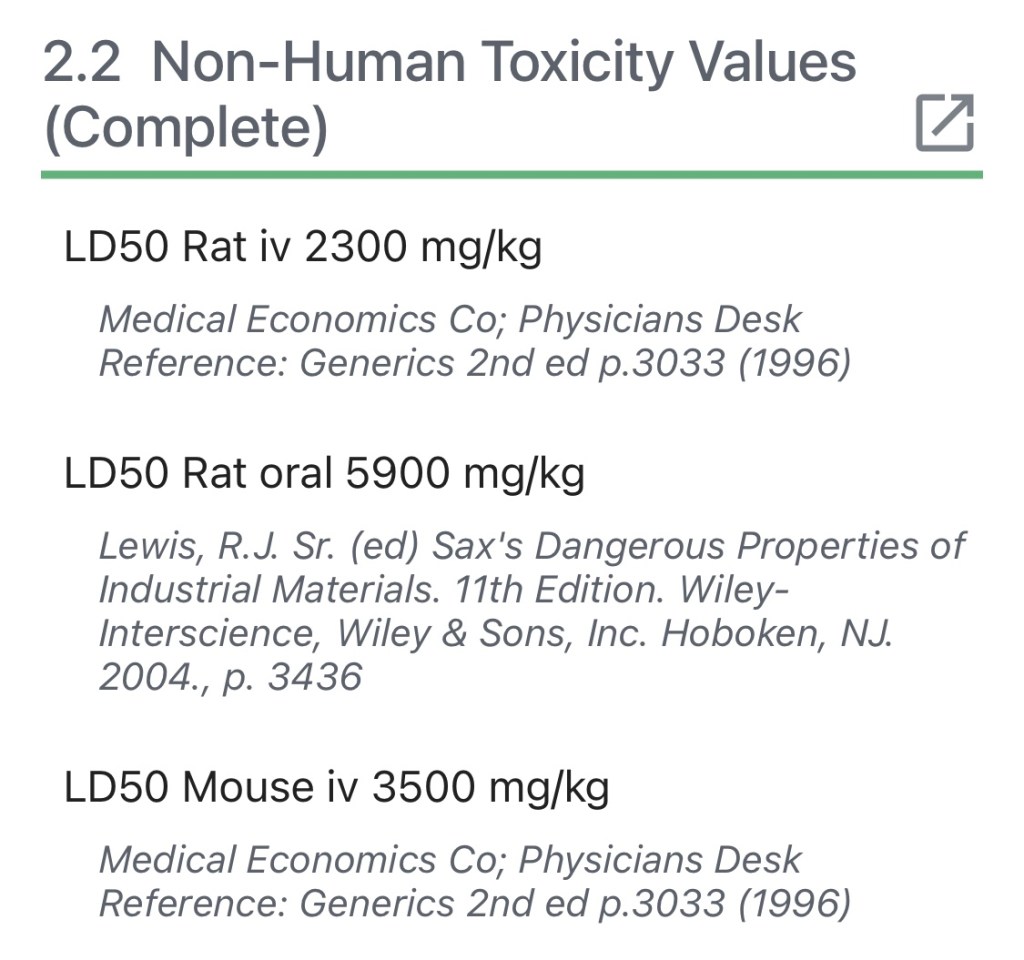
In addition, Tromethamine is known to cause increased blood coagulation time in dogs. As stated by the Nation Institutes of Health, “the possibility of such an occurrence in humans should be considered”.

Who manufactures Tromethamine?
Trometamine is manufactured for use in coronavirus vaccines through Pfizer and their suppliers. While the vaccines are manufactured by Pfizer, there are many international manufacturers that deliver cheap alternatives to domestically produced tromethamine. The FDA is financially unable to inspect all international manufacturing facilities. Could the substance be supplied by these facilities?
Is tromethamine supplied to Pfizer by an international supplier, (such as China) and what are the conditions of the facilities that make the substance? Could the accessibility, and low cost of tromethamine be enough to overlook its manufacturering conditions and potential toxicity?

Tromethamine FDA Recalls
The Comirnaty vaccine does not contain preservatives. Vaccines containing tromethamine have been recalled numerous times in the past by the FDA due to “particulate matter”, “potential presence of small particulates”, and “lack of sterility assurance”.
- Fresenius Kabi Issues Voluntary Nationwide Recall of 13 Lots of Ketorolac Tromethamine Injection, USP Due to the Presence of Particulate Matter in Reserve Samples
- Hikma Pharmaceuticals USA Inc. Extends Voluntary Nationwide Recall of Ketorolac Tromethamine Injection, USP 30mg/mL, 1mL Fill/2mL Vials Due to the Potential Presence of Small Particulates
- Sagent Pharmaceuticals Issues Voluntary Nationwide Recall of Ketorolac Tromethamine Injection, USP, 60mg/2mL (30mg per mL) Due to Lack of Sterility Assurance

Historically, there have been many instances of contamination in Chinese manufactured tromethamine. Beyond the substance’s potential toxicity, should Americans be concerned with the conditions of Pfizer’s international manufacturing facilities, and the quality of the tromethamine that is used in our children’s COVID-19 vaccines?
Statements
FDA –
“This new formulation is also authorized for use in individuals 12 years of age and older,” FDA spokesperson Abigail Capobiano said in an email to The Associated Press.
Pfizer –
“To enable extended storage time an alternative buffer is being leveraged, known as a ‘tris buffer,’” said Pfizer spokesperson Jerica Pitts. “This allows the mRNA to resist being degraded for a longer period of time before administration.”
Stanford University –
“The Pfizer data are super good with kids,” said Dr. Holden Maecker, professor of microbiology and immunology at Stanford University. “There are essentially no side effects in their study and they enrolled 20-30% of the kids with comorbid conditions, so obesity, diabetes—they had it all covered.”
Reuters released an official “fact check” statement that the newly added ingredient was not added to counter it’s side effects. They did not mention its potential toxicity.
Conclusion
Should even trace amounts of experimental chemicals with known toxicity be used in emergency use authorization issued vaccines? Although there have been many documented accounts of injecting Tromethamine into children during clinical trials, and the substance has been used in medicines, this will be the first time ever that tromethamine is mandated for use on a mass level to be injected into children aged five to eleven years old, no questions asked.
Should the future generations of our nation be subjected to routine tromethamine-laced injections, in accordance with federal and state regulations? Who decided upon the specific additives used and manufactured for COVID-19 vaccines? Could the decision to use tromethamine in COVID-19 vaccines be financially incentivized, regardless of potential chemical toxicity? What other substance could be used as a buffer in Comirnaty, instead of tromethamine?
Sources
- https://www.fda.gov/news-events/press-announcements/fda-authorizes-pfizer-biontech-covid-19-vaccine-emergency-use-children-5-through-11-years-age
- https://www.fda.gov/media/153717/download
- https://www.fda.gov/media/153447/download
- https://pubchem.ncbi.nlm.nih.gov/compound/Tromethamine#section=Safety-and-Hazards
- https://pubchem.ncbi.nlm.nih.gov/source/hsdb/3408#section=Non-Human-Toxicity-Excerpts-(Complete)
- https://www.drugs.com/sfx/tromethamine-side-effects.html
- https://www.genome.gov/10001345/importance-of-mouse-genome
- https://apnews.com/article/020421619563
- https://www.fda.gov/safety/recalls-market-withdrawals-safety-alerts/fresenius-kabi-issues-voluntary-nationwide-recall-13-lots-ketorolac-tromethamine-injection-usp-due
- https://www.fda.gov/safety/recalls-market-withdrawals-safety-alerts/hikma-pharmaceuticals-usa-inc-extends-voluntary-nationwide-recall-ketorolac-tromethamine-injection
- https://www.fda.gov/safety/recalls-market-withdrawals-safety-alerts/sagent-pharmaceuticals-issues-voluntary-nationwide-recall-ketorolac-tromethamine-injection-usp
- https://www.pfizer.com/sites/default/files/investors/financial_reports/annual_reports/2017/our-business-our-purpose/manufacturing-supply-chain-excellence/index.html
- https://www.google.com/amp/s/mobile.reuters.com/article/amp/idUSL1N2S31ML
- https://www.made-in-china.com/manufacturers/tromethamine.html
- https://www.pharmamanufacturing.com/industrynews/2010/052/