Just when the FDA could not contradict themselves any further, they have [unsurprisingly] succeeded yet again. The FDA has authorized the marketing of E-Cigarettes, while stating “it does not mean these products [E-Cigs] are safe or ‘FDA approved.’”
This announcement came while 2/3 of the mandated coronavirus injections are still under emergency use authorization along with most CoV-2 testing devices.
What is the message sent to the American people when the FDA grants authorization to market a product that is itself unapproved by the FDA, and even advised against by that same, and other, administrations? Should Americans blindly trust our national health institutions incentivized by financial profit?
“While today’s action permits the tobacco products to be sold in the U.S., it does not mean these products are safe or ‘FDA approved.’ All tobacco products are harmful and addictive and those who do not use tobacco products should not start.”

Controversy
On October 7th, 2021, the FDA posted a statement saying “The FDA has made a strong commitment and taken a number of actions to address the addictiveness posed by nicotine in cigarettes. However, there are now new concerns about the dangers from aerosols inhaled by users of e-cigarettes.”
“We see potential opportunity from the ENDS products for currently addicted adult smokers to help them quit smoking. But we also see risk, especially when it comes to youth use of these products. It’s important that we help answer difficult questions surrounding the dangers posed by aerosol from e-cigarette products. The data will help the agency as it continues to take appropriate action and advance policy steps to regulate these products.”
“Today, [October 7th, 2021] the U.S. Food and Drug Administration issued warning letters to 20 companies for continuing to unlawfully market electronic nicotine delivery system (ENDS) products that are the subject of Marketing Denial Orders (MDOs). These are the first warning letters issued for products subject to MDO determinations on their premarket tobacco product applications (PMTAs).”
October 12th, 2021 –
On October 12, 2021, just five days later, the FDA announced, e-cigs were now safe to market to the public. Although the FDA has not approved e-cigs, the marketing of e-cigs is fair game. To the FDA, the benefits of e-cigarettes marketing outweigh the long term public health risks.
“Today’s authorizations are an important step toward ensuring all new tobacco products undergo the FDA’s robust, scientific premarket evaluation. The manufacturer’s data demonstrates its tobacco-flavored products could benefit addicted adult smokers who switch to these products – either completely or with a significant reduction in cigarette consumption – by reducing their exposure to harmful chemicals,” said Mitch Zeller, J.D., director of the FDA’s Center for Tobacco Products. “We must remain vigilant with this authorization and we will monitor the marketing of the products, including whether the company fails to comply with any regulatory requirements or if credible evidence emerges of significant use by individuals who did not previously use a tobacco product, including youth. We will take action as appropriate, including withdrawing the authorization.”
The CDC’s Propaganda Against E-Cigs




E-Cigarette Demographic
According to House of Commons Research Library in 2019, 33% of adults vape to stop smoking, while 11.5 of ex-smokers have returned to the habit, 10% of 11-18 year olds have tried vaping at least once. This data depicts 41 million vapers globally, with e-cigarettes banned in seven countries
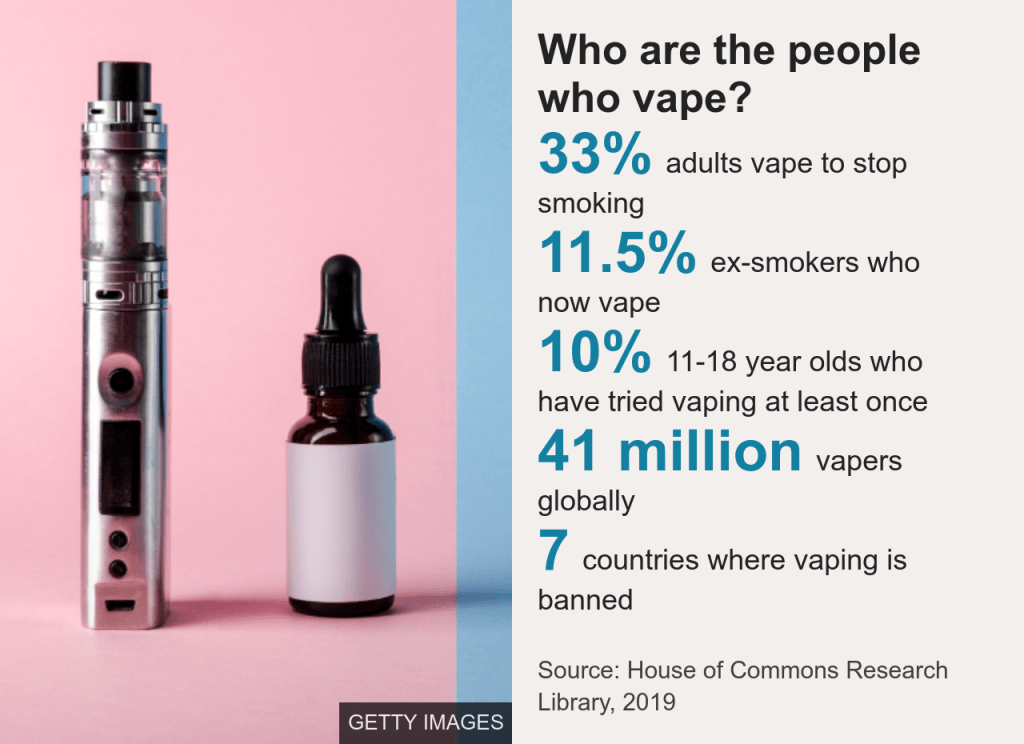

Data from the Truth Initiative in 2020 showed a deeper insight into the e-cigarette target demographic.

As of 2020, twenty-two countries had banned e-cigarettes, while six countries had restrictions set in place, including America.

Data from globaltobaccocontrol.org states that “As of February 2020, we have identified 100 countries that regulate or ban e-cigarettes.”

National Institutes of Health
The National Institutes of Health studied research on the effects of nanoparticles in e-cigarettes. Their findings are frightening.
“E-cigarettes generate high concentrations of nanoparticles and their chemical content requires further investigation.”
“Despite the small mass of nanoparticles, their toxicological impact could be significant.”
“Toxic chemicals that are attached to the small nanoparticles may have greater adverse health effects than when attached to submicron particles.”

“The presence of chromium, nickel, and lead, as well as tin, silver, and aluminum, was reported in e-cigarette aerosols, including metal nanoparticles. Lead and chromium concentrations were within the range of conventional cigarettes, whereas nickel was 2-100 times higher in e-cigarette aerosol than in Marlboro brand cigarette smoke.”
“Inhaled lead causes adverse health effects and elemental chromium is a respiratory irritant, while the hexavalent chromium that could be formed during high-temperature oxidation is a known human carcinogen.”

World Health Organization

Yet the FDA states that “the benefits” of marketing these products “outweigh the risks” when it comes to the marketing of these products. Was this same marketing perspective applied to the EUA of COVID-19 vaccines?
Conclusion
With almost half of e-cigarettes consumer demographic is children, how will the FDA’s approval of marketing e-cigarette products effect the future generations of America? Could increased use of e-cigarette nanoparticles have a greater ecotoxicological effect than traditional cigarette smoke? Should the FDA approve the marketing of an unapproved product, and how do these ethics apply to the “FDA approval” of modern medicine?

